Search
- Page Path
- HOME > Search
Original Articles
- Calcium & Bone Metabolism
- Decreased Serum Level of Sclerostin in Older Adults with Sarcopenia
- Seong Hee Ahn, Hee-Won Jung, Eunju Lee, Ji Yeon Baek, Il-Young Jang, So Jeong Park, Jin Young Lee, Eunah Choi, Yun Sun Lee, Seongbin Hong, Beom-Jun Kim
- Endocrinol Metab. 2022;37(3):487-496. Published online May 27, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1428

- 3,168 View
- 142 Download
- 11 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
Although muscles and bones interact with each other through various secretory factors, the role of sclerostin, an osteocyte-secreted factor, on muscle metabolism has not been well studied. We investigated the levels of serum sclerostin in Korean older adults with sarcopenia.
Methods
Blood samples were collected from 129 participants who underwent evaluation of muscle mass and function in an outpatient geriatric clinic of a teaching hospital. Sarcopenia and related parameters were determined using cutoff values for the Asian population. Serum sclerostin levels were measured using an enzyme-linked immunosorbent assay.
Results
The mean age of the participants was 69.6 years, and 20 participants (15.5%) were classified as having sarcopenia. After adjusting for age, sex, and body mass index, serum sclerostin levels were significantly lower in participants with sarcopenia, low muscle mass, or weak muscle strength (P=0.003 to 0.045). Serum sclerostin levels were positively associated with skeletal muscle index and grip strength after adjusting for confounders (P=0.001 and P=0.003), whereas sarcopenic phenotype score showed a negative association (P=0.006). These increases in muscle mass and strength were also dose dependent as serum sclerostin levels increased (P for trends=0.003 and P for trends=0.015). Higher serum sclerostin levels were associated with lower odds ratio (ORs) for sarcopenia, low muscle mass, and weak muscle strength after adjusting for confounders (OR, 0.27 to 0.50; P<0.001 to 0.025).
Conclusion
Higher serum sclerostin levels were associated with a lower risk of sarcopenia, low muscle mass, and weak muscle strength in Korean older adults. -
Citations
Citations to this article as recorded by- Mechanism and physical activities in bone-skeletal muscle crosstalk
Zhonghan Zhao, Kai Yan, Qiao Guan, Qiang Guo, Can Zhao
Frontiers in Endocrinology.2024;[Epub] CrossRef - Musculoskeletal disorders and coronary artery disease —promising molecular markers: literature review
Viktoria N. Karetnikova, Anastasiya G. Neeshpapa, Evgenia I. Carpova, Olga L. Barbarash
CardioSomatics.2024; 15(1): 55. CrossRef - Determinants of bone mass in older adults with normal- and overweight derived from the crosstalk with muscle and adipose tissue
Carina O. Walowski, Catrin Herpich, Janna Enderle, Wiebke Braun, Marcus Both, Mario Hasler, Manfred J. Müller, Kristina Norman, Anja Bosy-Westphal
Scientific Reports.2023;[Epub] CrossRef - Role of the Osteocyte in Musculoskeletal Disease
Anika Shimonty, Lynda F. Bonewald, Fabrizio Pin
Current Osteoporosis Reports.2023; 21(3): 303. CrossRef - The role of sclerostin in lipid and glucose metabolism disorders
Hewen Jiang, Dijie Li, Ying Han, Nanxi Li, Xiaohui Tao, Jin Liu, Zongkang Zhang, Yuanyuan Yu, Luyao Wang, Sifan Yu, Ning Zhang, Huan Xiao, Xin Yang, Yihao Zhang, Ge Zhang, Bao-Ting Zhang
Biochemical Pharmacology.2023; 215: 115694. CrossRef - Cytokines and exosomal miRNAs in skeletal muscle–adipose crosstalk
Liu Guo, Menchus Quan, Weijun Pang, Yulong Yin, Fengna Li
Trends in Endocrinology & Metabolism.2023; 34(10): 666. CrossRef - Sclerostin: clinical insights in muscle–bone crosstalk
Antimo Moretti, Giovanni Iolascon
Journal of International Medical Research.2023;[Epub] CrossRef - Anti-sclerostin antibodies: a new frontier in fragility fractures treatment
Giovanni Iolascon, Sara Liguori, Marco Paoletta, Giuseppe Toro, Antimo Moretti
Therapeutic Advances in Musculoskeletal Disease.2023;[Epub] CrossRef - Sclerostin as a Putative Myokine in Sarcopenia
Hyon-Seung Yi
Endocrinology and Metabolism.2022; 37(3): 430. CrossRef - Organokines, Sarcopenia, and Metabolic Repercussions: The Vicious Cycle and the Interplay with Exercise
Giulia Minniti, Letícia Maria Pescinini-Salzedas, Guilherme Almeida dos Santos Minniti, Lucas Fornari Laurindo, Sandra Maria Barbalho, Renata Vargas Sinatora, Lance Alan Sloan, Rafael Santos de Argollo Haber, Adriano Cressoni Araújo, Karina Quesada, Jesse
International Journal of Molecular Sciences.2022; 23(21): 13452. CrossRef
- Mechanism and physical activities in bone-skeletal muscle crosstalk

- Adrenal Gland
- Aldosterone Inhibits In Vitro Myogenesis by Increasing Intracellular Oxidative Stress via Mineralocorticoid Receptor
- Jin Young Lee, Da Ae Kim, Eunah Choi, Yun Sun Lee, So Jeong Park, Beom-Jun Kim
- Endocrinol Metab. 2021;36(4):865-874. Published online July 30, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1108
- 4,301 View
- 112 Download
- 7 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
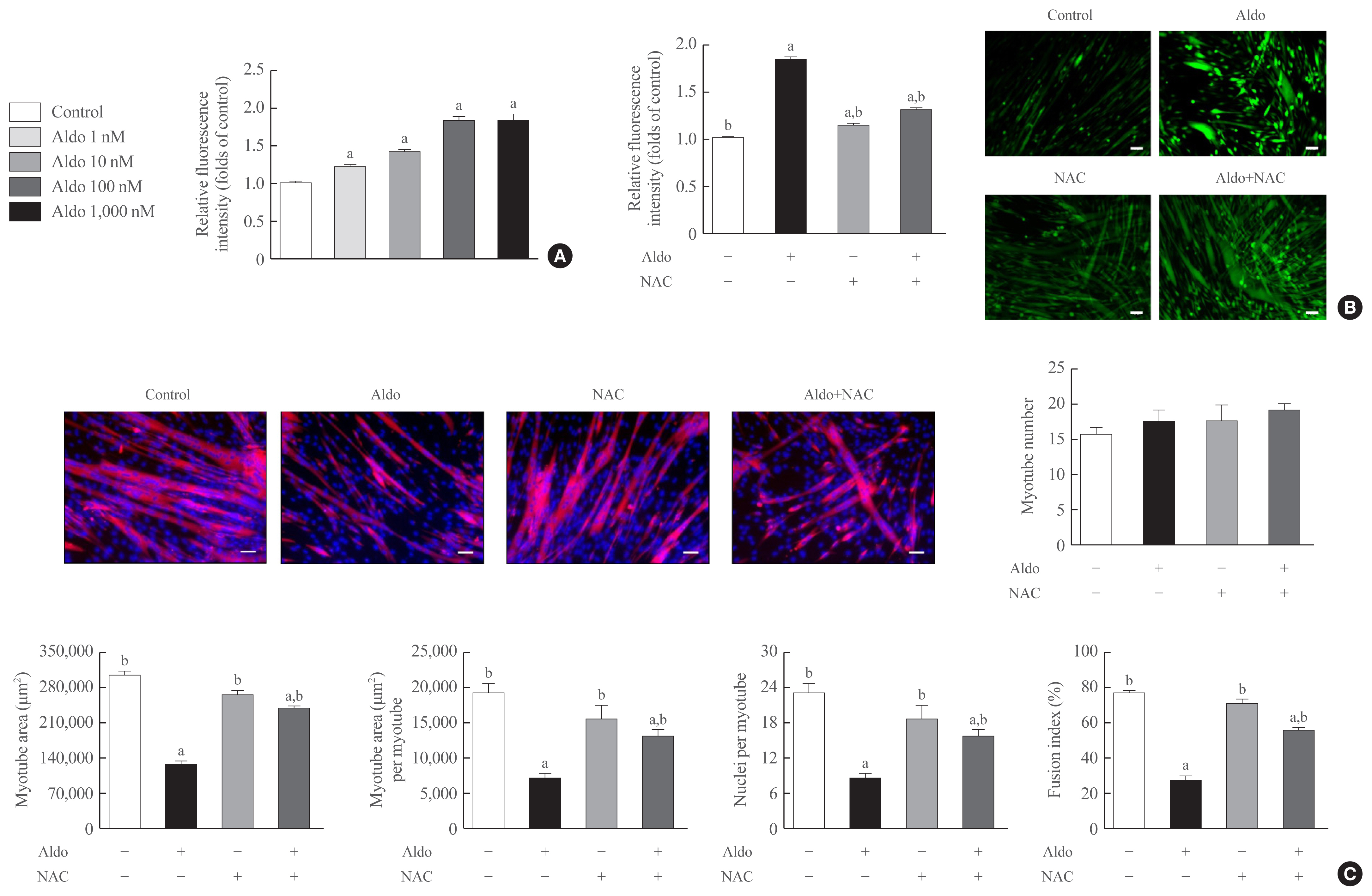
Despite clinical evidence indicating poor muscle health in subjects with primary aldosteronism (PA), it is still unclear whether the role of aldosterone in muscle metabolism is direct or mediated indirectly via factors, such as electrolyte imbalance or impaired glucose uptake. As one approach to clarify this issue, we investigated the effect of aldosterone on in vitro myogenesis and the potential mechanism explaining it.
Methods
Myogenesis was induced in mouse C2C12 myoblasts with 2% horse serum. Immunofluorescence, quantitative reversetranscription polymerase chain reaction, Western blot, viability, and migration analyses were performed for experimental research.
Results
Recombinant aldosterone treatment suppressed muscle differentiation from mouse C2C12 myoblasts in a dose-dependent manner, and consistently reduced the expression of myogenic differentiation markers. Furthermore, aldosterone significantly increased intracellular reactive oxygen species (ROS) levels in myotubes, and treatment with N-acetyl cysteine, a potent biological thiol antioxidant, reversed the decrease of myotube area, myotube area per myotube, nucleus number per myotube, and fusion index due to aldosterone through decreasing oxidative stress. A binding enzyme-linked immunosorbent assay confirmed that mineralocorticoid receptor (MR) interacted with aldosterone in C2C12 myoblasts, while eplerenone, an MR inhibitor, blocked aldosterone-stimulated intracellular ROS generation during myogenesis and markedly attenuated the suppression of in vitro myogenesis by aldosterone.
Conclusion
These findings support the hypothesis that hypersecretion of aldosterone, like PA, directly contributes to muscular deterioration and suggest that antioxidants and/or MR antagonists could be effective therapeutic options to reduce the risk of sarcopenia in these patients. -
Citations
Citations to this article as recorded by- Molecular mechanisms underlying sarcopenia in heart failure
Cody A. Rutledge
The Journal of Cardiovascular Aging.2024;[Epub] CrossRef - Role of glucocorticoid and mineralocorticoid receptors in rainbow trout (Oncorhynchus mykiss) skeletal muscle: A transcriptomic perspective of cortisol action
Jorge E. Aedo, Rodrigo Zuloaga, Daniela Aravena-Canales, Alfredo Molina, Juan Antonio Valdés
Frontiers in Physiology.2023;[Epub] CrossRef - Effect of 11-Deoxycorticosterone in the Transcriptomic Response to Stress in Rainbow Trout Skeletal Muscle
Rodrigo Zuloaga, Daniela Aravena-Canales, Jorge Eduardo Aedo, Cesar Osorio-Fuentealba, Alfredo Molina, Juan Antonio Valdés
Genes.2023; 14(2): 512. CrossRef - Heart Failure Medication and Muscle Wasting
Yasuhiro Izumiya
Circulation Journal.2023; 88(1): 20. CrossRef - 2023 Korean Endocrine Society Consensus Guidelines for the Diagnosis and Management of Primary Aldosteronism
Jeonghoon Ha, Jung Hwan Park, Kyoung Jin Kim, Jung Hee Kim, Kyong Yeun Jung, Jeongmin Lee, Jong Han Choi, Seung Hun Lee, Namki Hong, Jung Soo Lim, Byung Kwan Park, Jung-Han Kim, Kyeong Cheon Jung, Jooyoung Cho, Mi-kyung Kim, Choon Hee Chung
Endocrinology and Metabolism.2023; 38(6): 597. CrossRef - Higher Plasma Stromal Cell-Derived Factor 1 Is Associated with Lower Risk for Sarcopenia in Older Asian Adults
Sunghwan Ji, Kyunggon Kim, So Jeong Park, Jin Young Lee, Hee-Won Jung, Hyun Ju Yoo, Il-Young Jang, Eunju Lee, Ji Yeon Baek, Beom-Jun Kim
Endocrinology and Metabolism.2023; 38(6): 701. CrossRef - The Role of Aldosterone in OSA and OSA-Related Hypertension
Yi Wang, Chuan Xiang Li, Ying Ni Lin, Li Yue Zhang, Shi Qi Li, Liu Zhang, Ya Ru Yan, Fang Ying Lu, Ning Li, Qing Yun Li
Frontiers in Endocrinology.2022;[Epub] CrossRef - Mineralocorticoid Receptor Antagonists in Diabetic Kidney Disease
Daiji Kawanami, Yuichi Takashi, Yoshimi Muta, Naoki Oda, Dai Nagata, Hiroyuki Takahashi, Makito Tanabe
Frontiers in Pharmacology.2021;[Epub] CrossRef
- Molecular mechanisms underlying sarcopenia in heart failure


 KES
KES

 First
First Prev
Prev



